Electrochemical production of active chlorine using platinised platinum electrodes
Main Article Content
Abstract
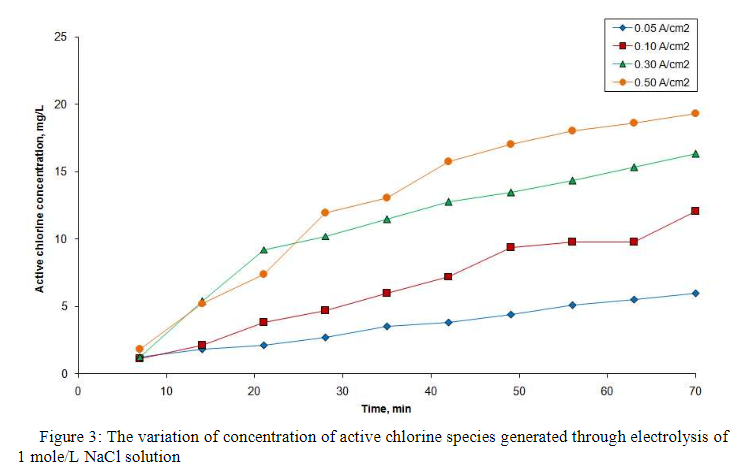
This paper presents the results obtained during the electrogeneration of active chlorine species. Active chlorine species were generated through the electrolysis of sodium chloride solutions in an undivided cell, employing two platinised platinum electrodes and a polarity reversal procedure. The electrolytic process was conducted at four current densities (0.05, 0.10, 0.30 and 0.50 A/cm2) and three concentrations (0.1, 0.5 and1 mole/L) the polarity reversal period being of 5 min. The brine flow rate was kept constant, namely 1.68 L/h. The results showed a maximum active chlorine concentrations for an initial brine concentration of 0.5 mole/L. The overall concentration of dissolved chlorine in water was quantified as active chlorine, defined as the sum of the three possible species (Cl2, HClO and ClO-).